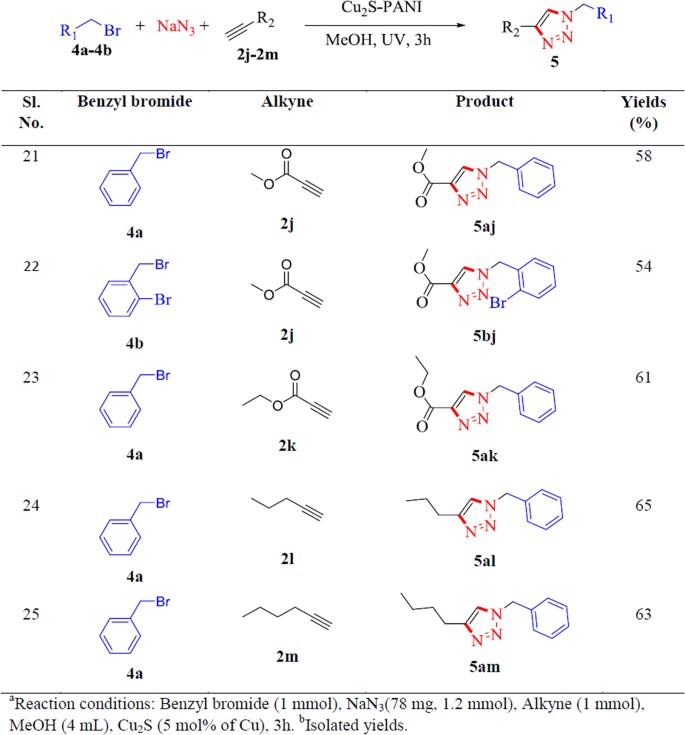
![Visible Light Assisted Photocatalytic [3 + 2] Azide–Alkyne “Click” Reaction for the Synthesis of 1,4-Substituted 1,2,3-Triazoles Using a Novel Bimetallic Ru–Mn Complex | ACS Sustainable Chemistry & Engineering Visible Light Assisted Photocatalytic [3 + 2] Azide–Alkyne “Click” Reaction for the Synthesis of 1,4-Substituted 1,2,3-Triazoles Using a Novel Bimetallic Ru–Mn Complex | ACS Sustainable Chemistry & Engineering](https://pubs.acs.org/cms/10.1021/acssuschemeng.5b00653/asset/images/medium/sc-2015-00653z_0003.gif)
Visible Light Assisted Photocatalytic [3 + 2] Azide–Alkyne “Click” Reaction for the Synthesis of 1,4-Substituted 1,2,3-Triazoles Using a Novel Bimetallic Ru–Mn Complex | ACS Sustainable Chemistry & Engineering
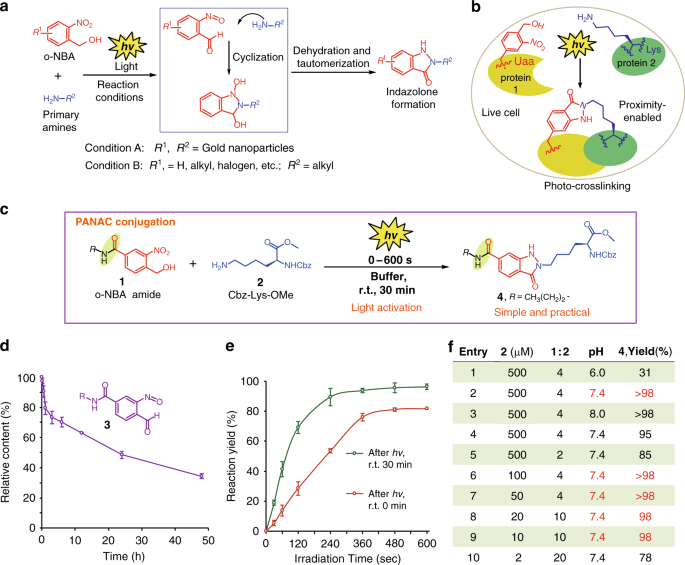
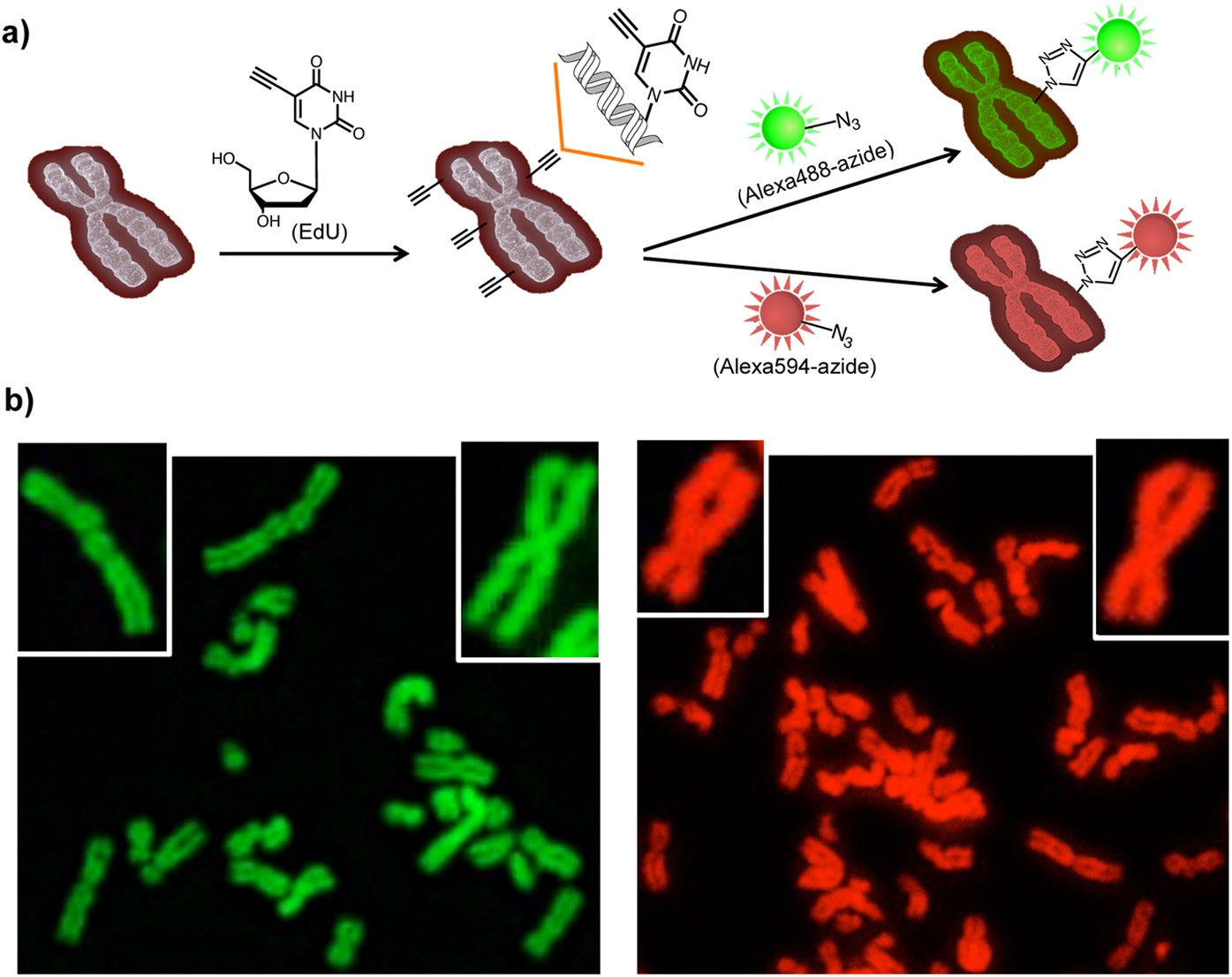
Light-induced primary amines and o-nitrobenzyl alcohols cyclization as a versatile photoclick reaction for modular conjugation | Nature Communications

Light‐Induced Click Reactions - Tasdelen - 2013 - Angewandte Chemie International Edition - Wiley Online Library
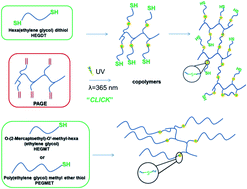
Thiol–ene coupling reaction achievement and monitoring by “in situ” UV irradiation NMR spectroscopy - RSC Advances (RSC Publishing)
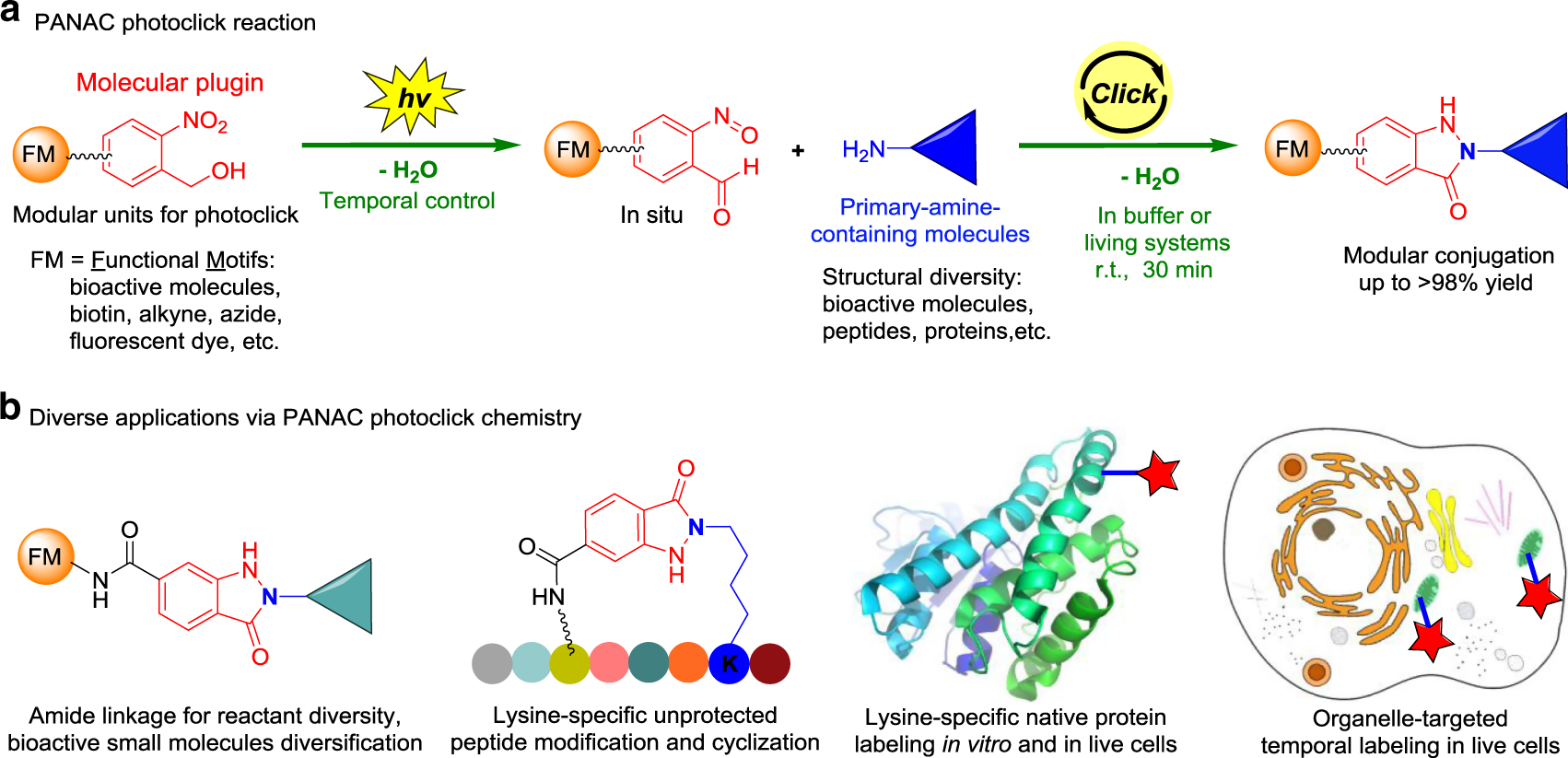
Light-induced primary amines and o-nitrobenzyl alcohols cyclization as a versatile photoclick reaction for modular conjugation | Nature Communications
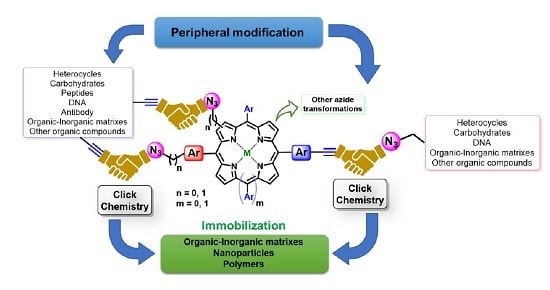
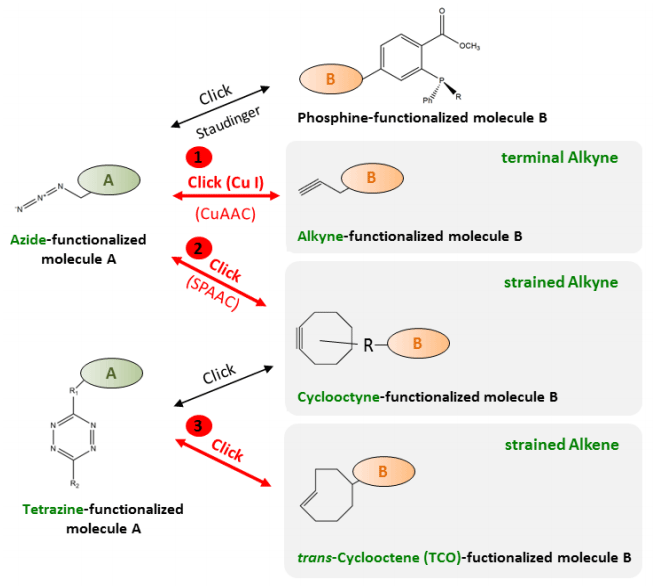
Molecules | Free Full-Text | Azides and Porphyrinoids: Synthetic Approaches and Applications. Part 1—Azides, Porphyrins and Corroles
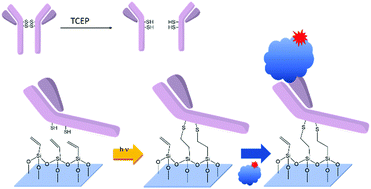
Thiol–ene click chemistry towards easy microarraying of half-antibodies - Chemical Communications (RSC Publishing)

Photoinitiated Alkyne–Azide Click and Radical Cross-Linking Reactions for the Patterning of PEG Hydrogels | Biomacromolecules

Influence of Type of Initiation on Thiol–Ene “Click” Chemistry - Uygun - 2010 - Macromolecular Chemistry and Physics - Wiley Online Library
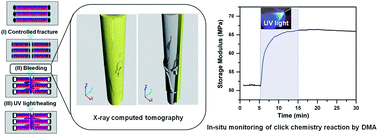
Real time monitoring of click chemistry self-healing in polymer composites - Journal of Materials Chemistry A (RSC Publishing)
![Direct ring-strain loading for visible-light accelerated bioorthogonal ligation via diarylsydnone-dibenzo[b,f ][1,4,5]thiadiazepine photo-click reactions | Communications Chemistry Direct ring-strain loading for visible-light accelerated bioorthogonal ligation via diarylsydnone-dibenzo[b,f ][1,4,5]thiadiazepine photo-click reactions | Communications Chemistry](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs42004-020-0273-6/MediaObjects/42004_2020_273_Fig1_HTML.png)
Direct ring-strain loading for visible-light accelerated bioorthogonal ligation via diarylsydnone-dibenzo[b,f ][1,4,5]thiadiazepine photo-click reactions | Communications Chemistry

Photocrosslinking and Click Chemistry Enable the Specific Detection of Proteins Interacting with Phospholipids at the Membrane Interface - ScienceDirect

Light‐Induced Click Reactions - Tasdelen - 2013 - Angewandte Chemie International Edition - Wiley Online Library